Treating the charges on nitrogen and argon as the sums of charges on protons, electrons and neutrons, it is deduced that the proton charge is (1±4×10 −20) e and the charge on the neutron is less than 4 × −20 e. Table 2.1 Charges and masses of the particles within atoms. We have found that the charge on an argon atom (18 protons, 18 electrons and 22 neutrons) is not greater than 8×10 −20 e and that on a nitrogen molecule (14 protons, 14 electrons and 14 neutrons) is not greater than 12 × 10 −20 e. Both protons and neutrons have a mass of 1, while electrons have almost no mass. In a nucleus, the protons and neutrons are stuck together. It therefore seemed desirable to find whether matter in which there is an excess of neutrons is electrically neutral. 20 For example, when we put two objects of +10C and +20C together and then take them apart, each of them acquires a charge of +15C. Since matter also contains neutrons, they have assumed in effect that the neutron has a charge equal to that of a hydrogen atom but the neutron might equally well have a charge opposite to that of a hydrogen atom, in which case their experiment does not settle the point at issue, since carbon dioxide contains equal numbers of protons and neutrons (to within 0.1 per cent). Charge Of Proton Proton, a stable subatomic particle that has a positive charge equal in magnitude to a unit of electron charge and a rest mass of 1.67262 × 10 27 kg, which is 1,836 times the mass of an electron. In fact, Piccard and Kessler 3 attempted to detect such a difference in 1925, and found that a molecule of carbon dioxide did not have an electric charge greater than 2×10 −19 e, where - e is the electronic charge, from which they concluded that the magnitudes of the proton and electron charges were the same to within 5 parts in 10 21, assuming that matter was built entirely of protons and electrons. IT has recently been suggested by Bondi and Ly ttleton 1,2 that the magnitudes of the electric charges on the proton and electron may differ by a little more than one part in 10 18, in which case electrostatic forces would cause the universe to expand. When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. It is an important input for bound-state Quantum Electrodynamic (QED) calculations of the hydrogen atomic energy levels. (i) Chemical reaction: Na +Cl )+(e NaCl (1.1) )0 (+ (ii) (iii) (iv) (v) Radioactive decay: A neutron will decay into a positively charged proton, a negatively charged electron and a neutral charged electron-anti-neutrino, n p + e + (1. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. The electric charge radius of a proton, denoted by, characterizes the spatial distribution of its electric charge carried by the quarks. If a proton is accelerated from rest through a potential difference of 30 kV, it acquires an energy of 30 keV. For example, about 5 eV of energy is required to break up certain organic molecules. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. To have a physical quantity that is independent of test charge, we define electric potential V (or simply potential, since electric is understood). If there are more electrons than protons, then the net charge on the object is negative. The mass of a proton is 1.007277 amu or 1.67252 X 10-27 kg. The rms-radius R of the proton charge distribution is a fundamental quantity needed for precision physics. If an object has more protons than electrons, then the net charge on the object is positive. Field PHY 2049 Chapter 22 chp222.  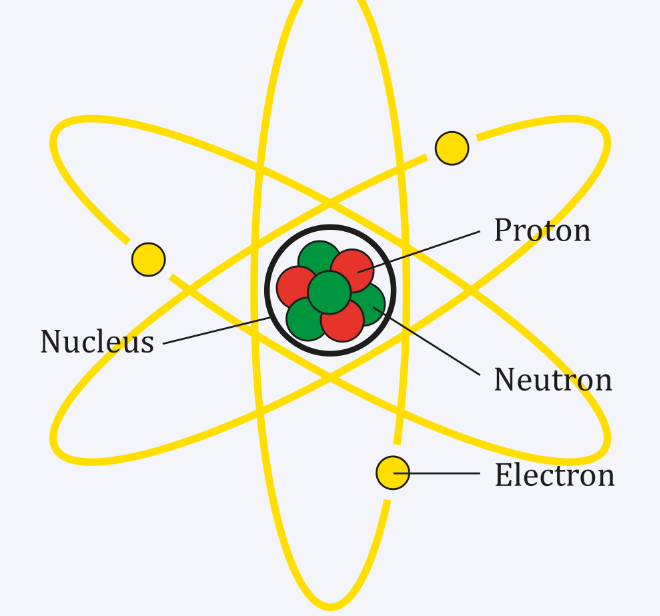
The charge is one positive fundamental unit, or 1.602 × 1019 coulomb. Electric Charge : electron charge -e e 1.6x10-19C proton charge e C Coulomb Electric charge is a conserved quantity (net electric charge is never created or destroyed ) q1q2 r R. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive. Proton: The positively charged fundamental particle present in the atom is called proton. The +1 represents the polarity and the quantity of the charge the proton carries.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
